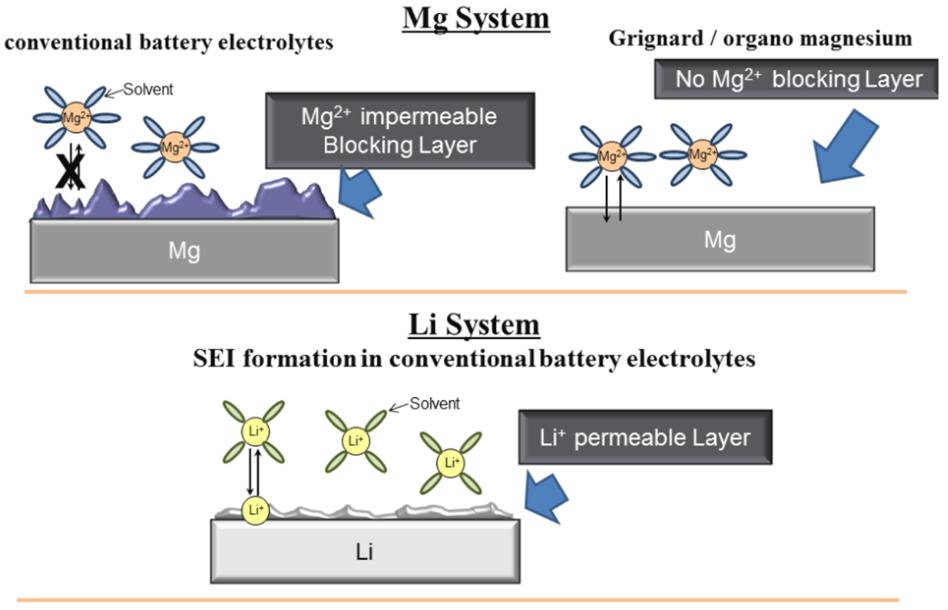
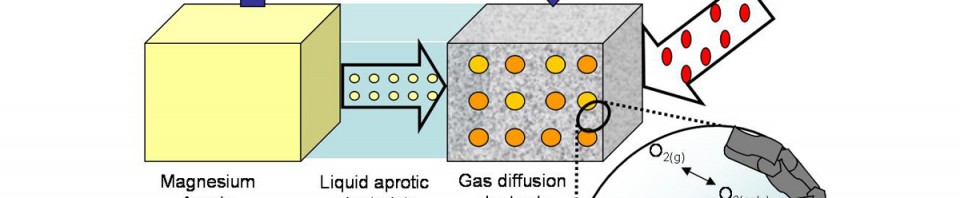
Magnesium batteries are promising next-generation energy-storage devices due to their high theoretical capacities and low raw-materials costs. Monolithic Mg negative electrodes also appear to have a smaller propensity for dendrite formation than those based on metallic lithium, making Mg a potentially safer alternative for long-lived, high-capacity batteries. Despite this promise, many challenges must be addressed at the materials level before Mg-batteries can achieve widespread commercialization. Two of these challenges relate to the development of efficient electrolytes and to the demonstration of rechargeable cells that approach theoretically expected energy densities.
This talk will summarize our efforts to address these issues using a combination of computational and experimental techniques [1-4]. Emphasis will be placed on understanding the performance of ‘Mg-air’ batteries and characterizing processes occurring within Mg-electrolytes, and at their interfaces with electrodes.
1. N. Kumar and D. J. Siegel, J. Phys. Chem. Lett., 7, 874-881 (2016).
2. J. G. Smith, J. Naruse, H. Hiramatsu, and D. J. Siegel, Chem. Mater., 28, 1390-1401 (2016).
3. G. Vardar et al., Chem. Mater., 27, 7564 (2015).
4. G. Vardar et al., ACS Appl. Materials & Interfaces 6, 18033 (2014).