Sodium-ion batteries may become an inexpensive alternative to lithium-ion batteries for large-scale stationary storage of energy generated by intermittent renewable sources. The key for the deployment of this technology is the development of suitable anode materials which can rival the graphite anodes used in lithium-ion batteries in terms of energy density, cycle life, rate performance, and safety.
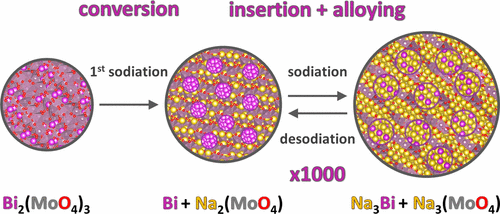
Here, we demonstrate that the bismuth metalates, BiVO4 and Bi2(MoO4)3, as representatives of ternary metalates, can cope with these requirements. High specific capacities (367 mAh/g and 352 mAh/g, respectively), exceptionally high cycling stability for alloying anodes (up to 79% of the first charge capacity is retained over 1000 cycles at ∼1C for Bi2(MoO4)3), better high-rate performance compared to other Bi-based anodes, low environmental load (Bi has low toxicity for a heavy metal), and low manufacturing costs (e.g., BiVO4 is a commercial yellow pigment) make this novel class of anode materials suitable for large-scale electrical energy storage applications. Operando XANES, ex situ XRD, and DFT analysis suggest that the initial compounds are converted into alloying Bi nanocrystallites confined in a matrix of electrochemically active insertion hosts Na3+xVO4 and Na2+xMoO4, respectively. The Bi metalate phases are not reformed on charge, and on subsequent cycles the reaction with Bi metal and vanadate/molybdate phases gives rise to the reversible capacity. The nanostructured composite anode thus obtained has excellent high rate performance and retains its capacity over hundreds of cycles.